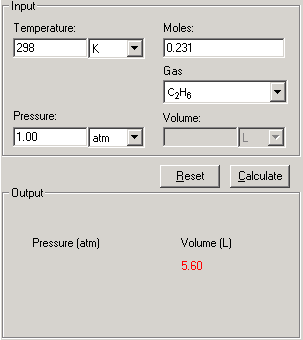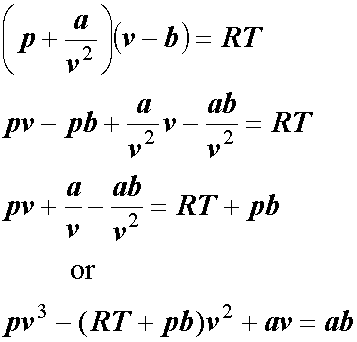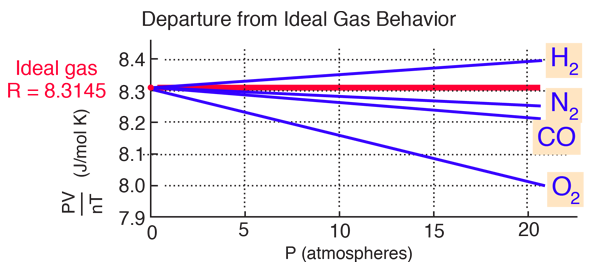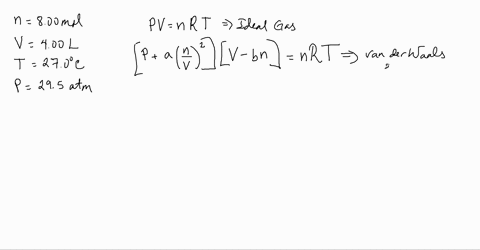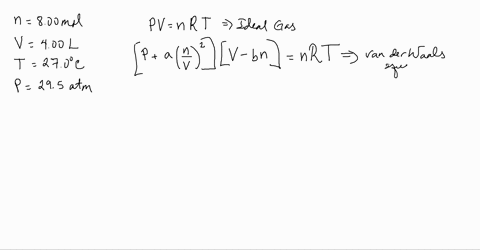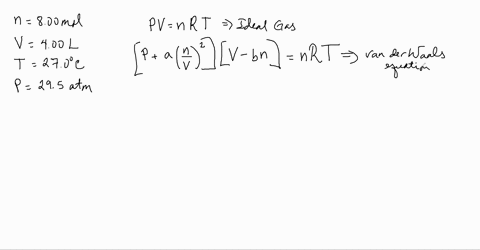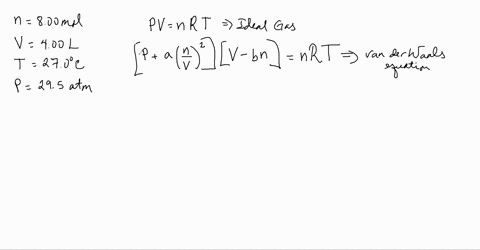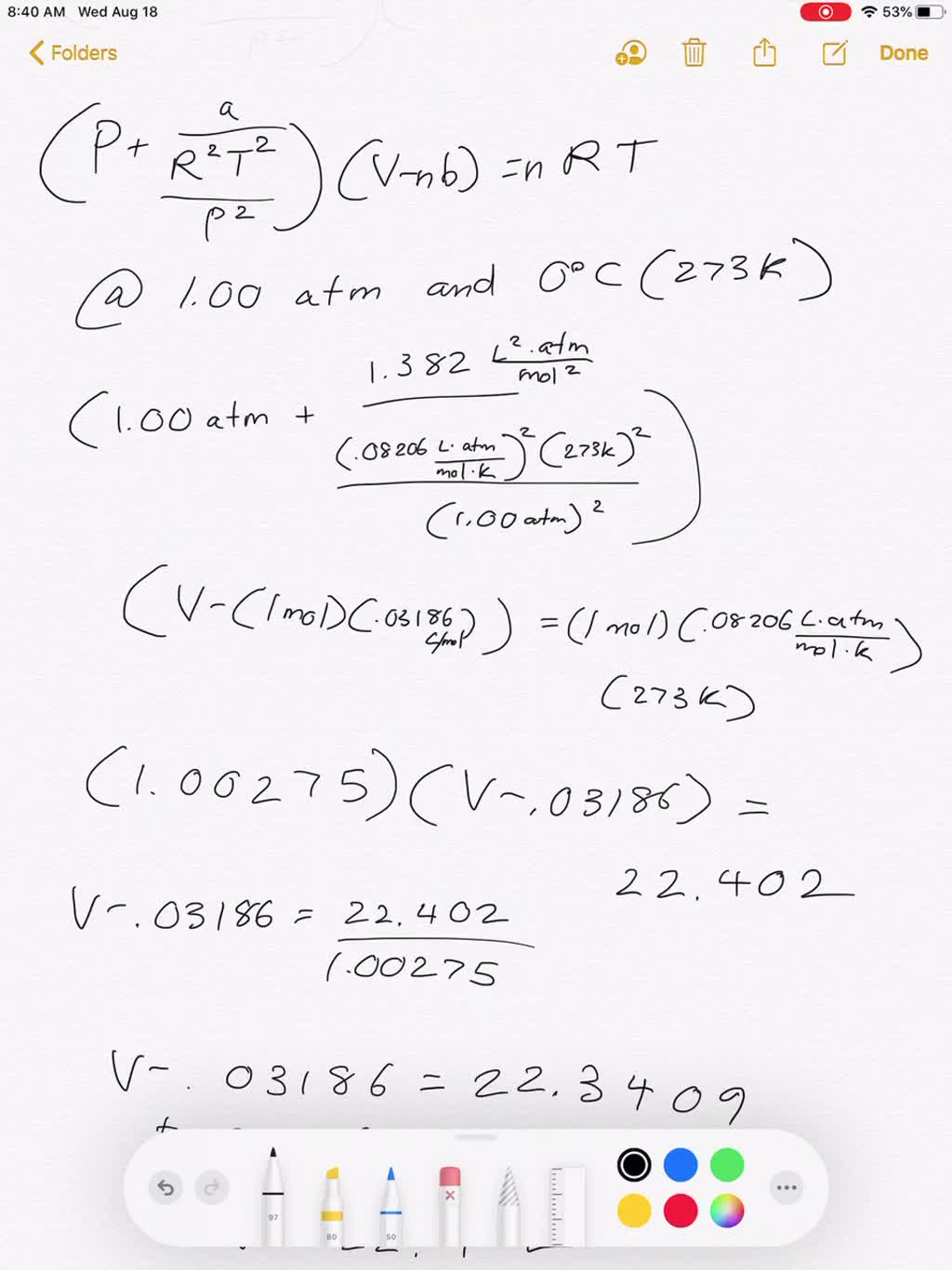
Use the Van der Waals Law Calculator to find the volume of an ideal gas, nitrogen, and oxygen. How close are the values? Do you feel that that treating air (which is 78% nitrogen and 21% oxygen) as an ...
Using Vander Waals equation calculate the pressure exerted by one mole of CO2. Its volume at 373 K is 0.05 dm^3 . - Sarthaks eConnect | Largest Online Education Community

Calculate the radius of He atoms if its van der Waal's constant 'b' is 24mL `"mol"^(-1)`. (Note: mL=
Using Vander Waal's equation, calculate the constant 'a' when two moles of a gas confined in a four litre flask exerts a pressure of 11.0 - Sarthaks eConnect | Largest Online Education Community

Using van der Waals equation, calculate the constant a when two moles of a gas confined in a four litre flask exert a pressure of 11.0 atm at a temperature of 300