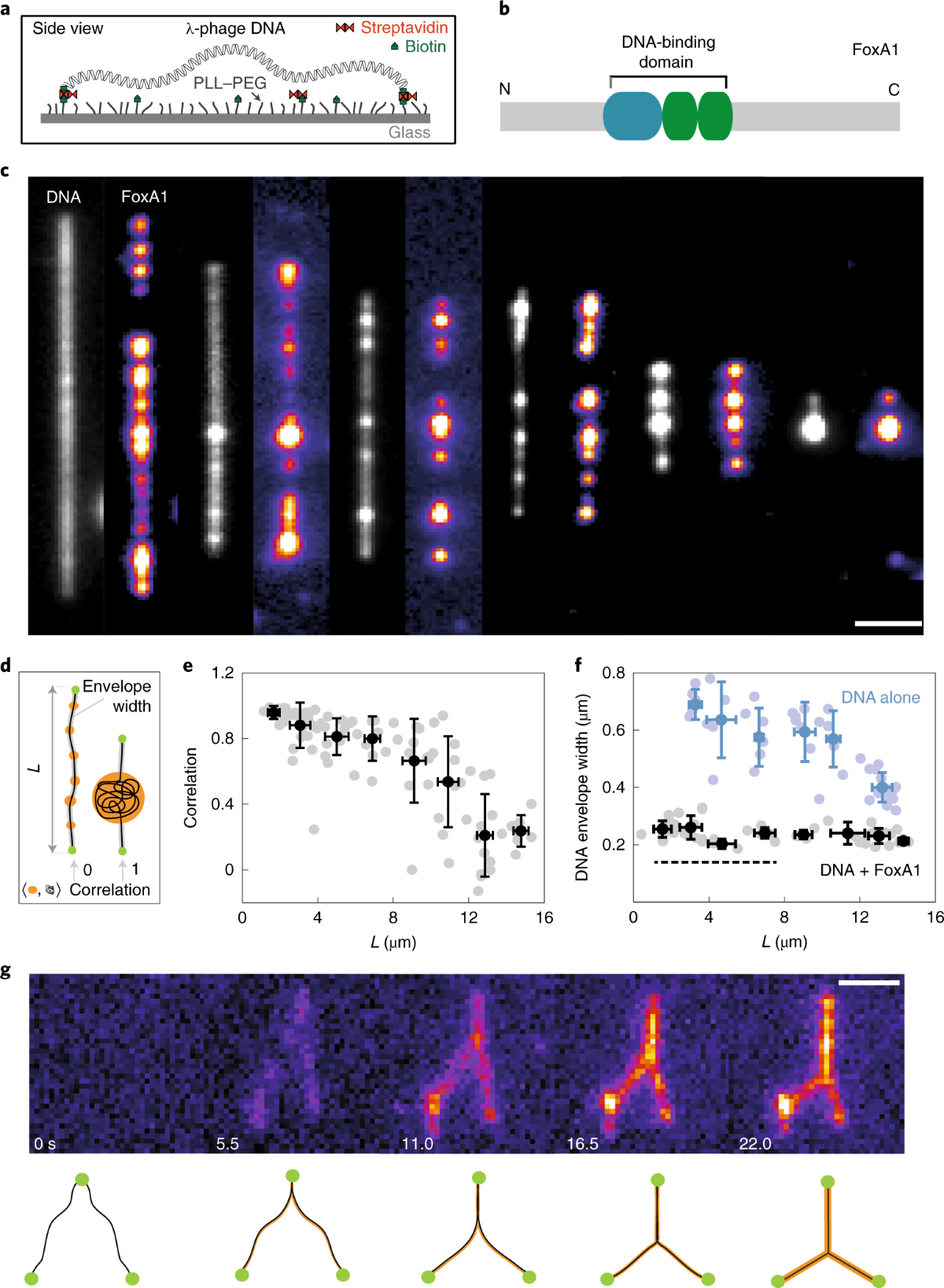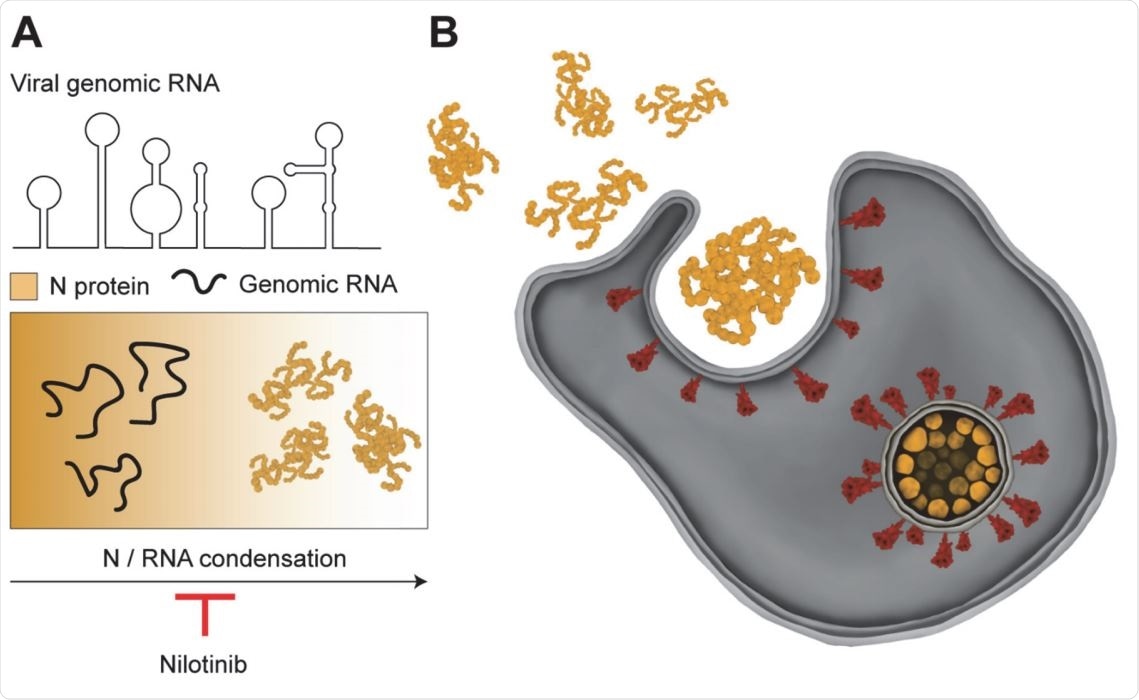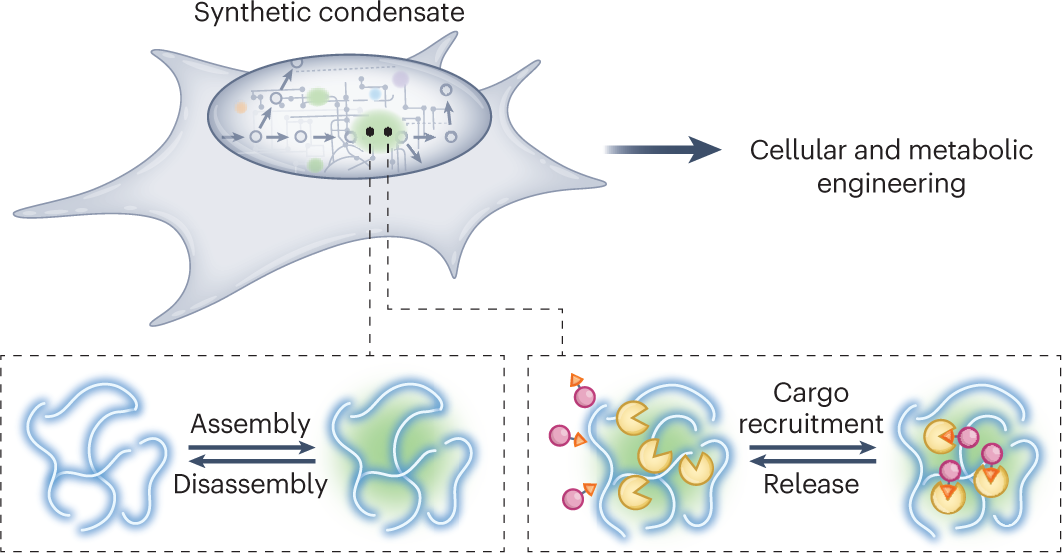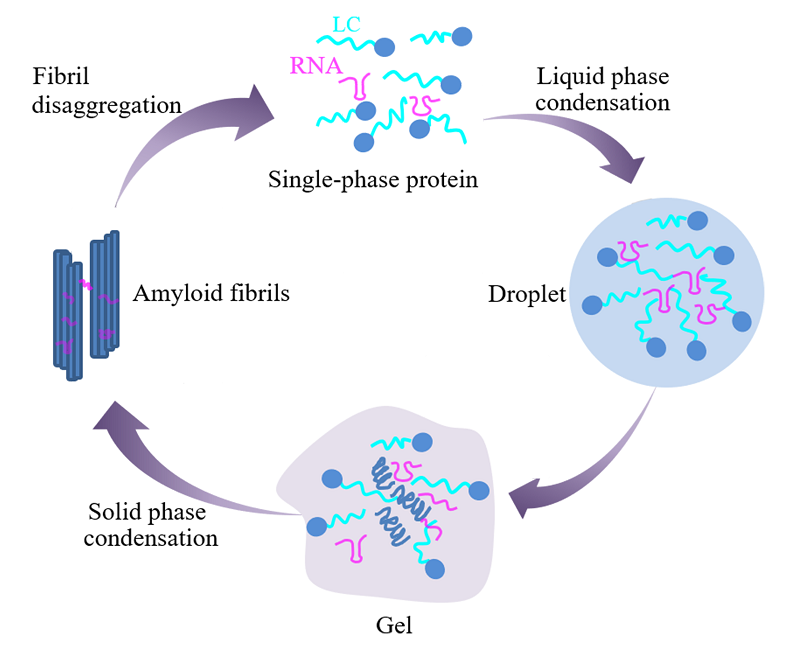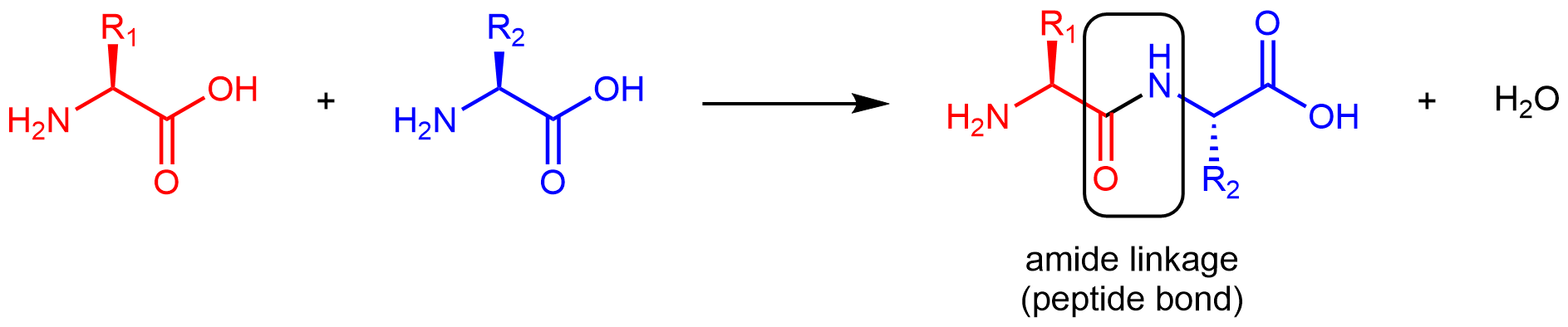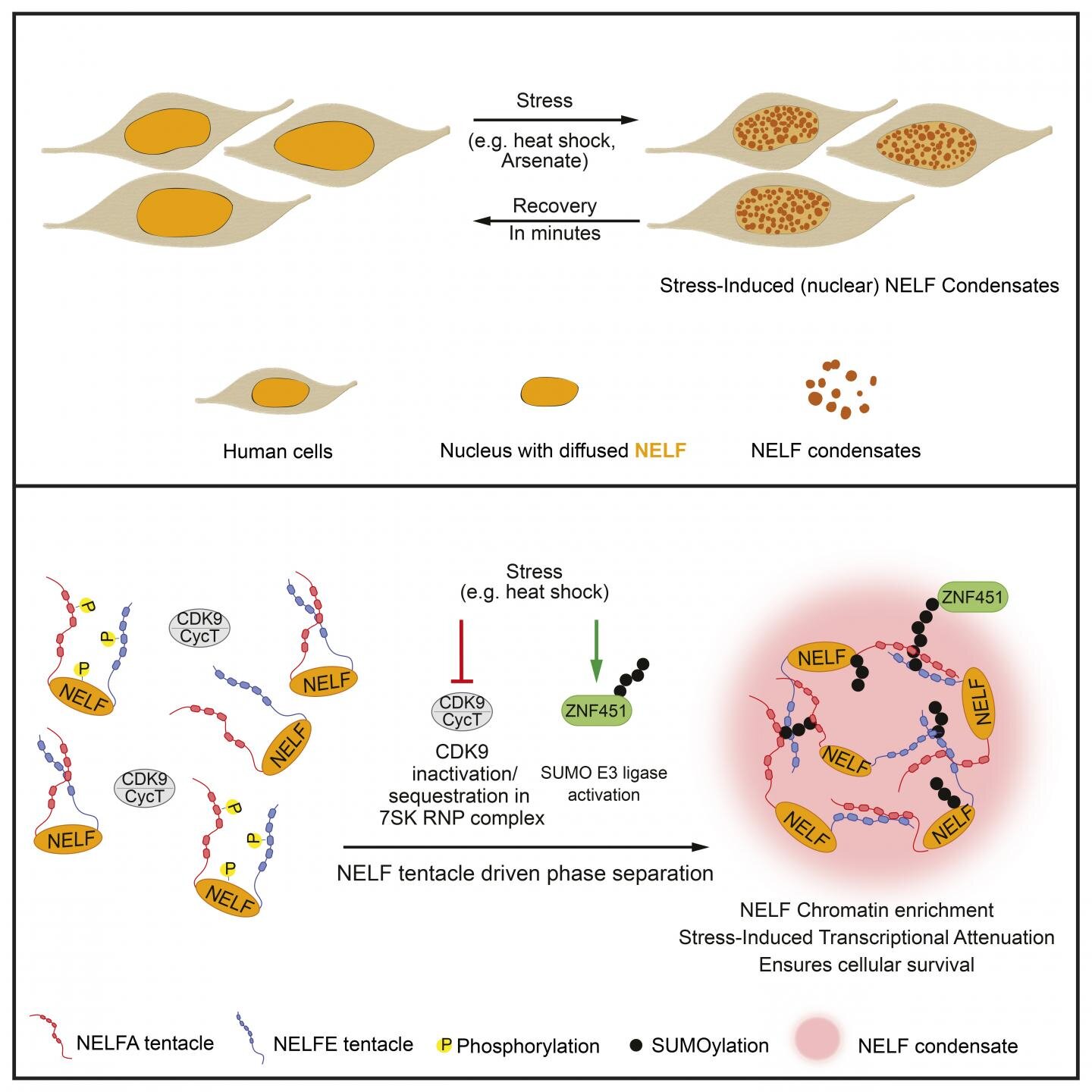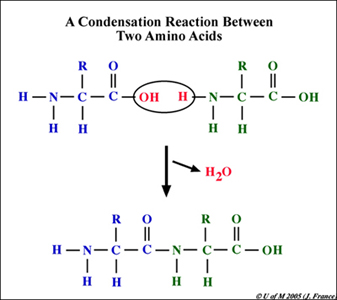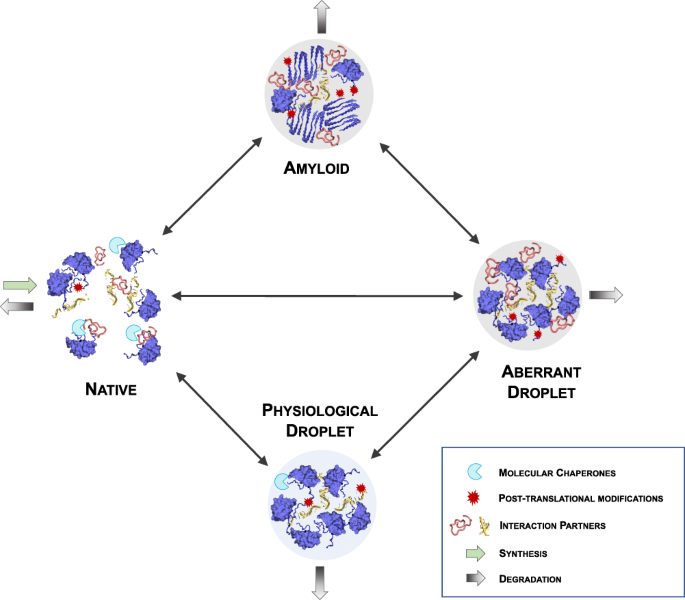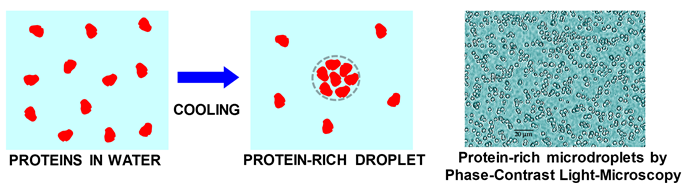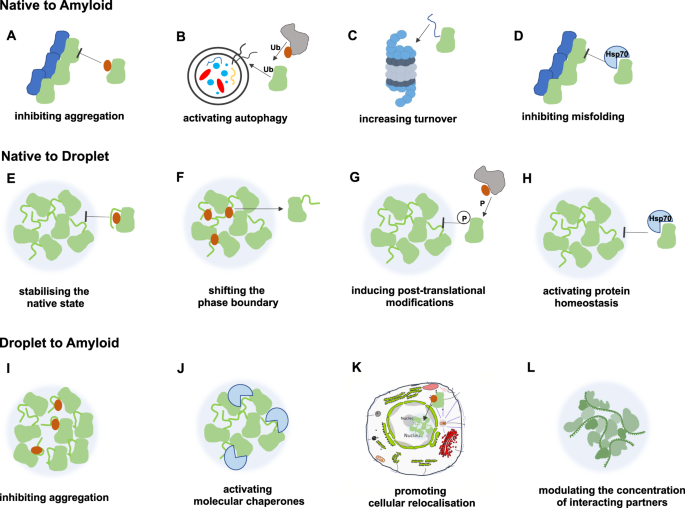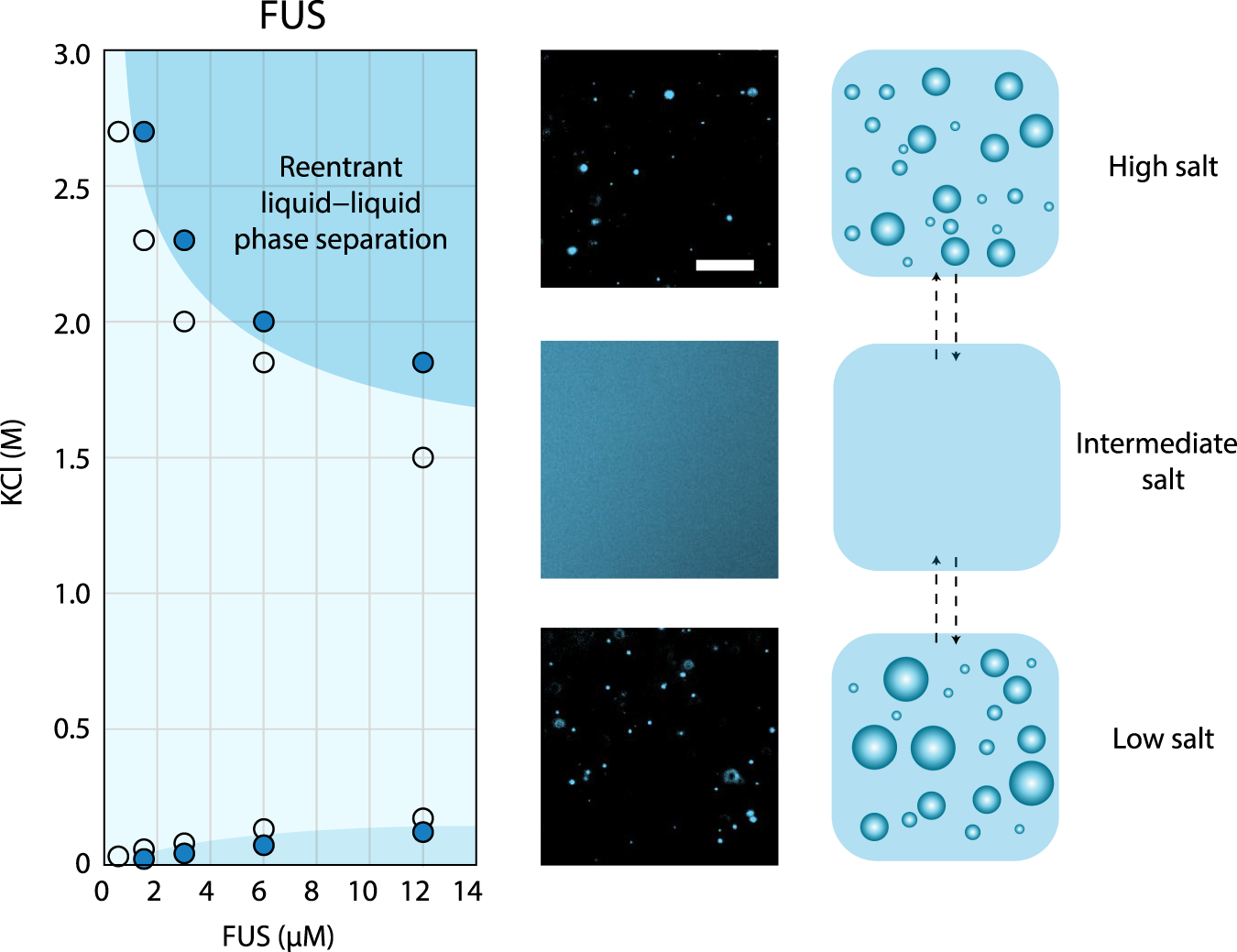
Reentrant liquid condensate phase of proteins is stabilized by hydrophobic and non-ionic interactions | Nature Communications

Condensation reaction on mineral surfaces, where activated monomers... | Download Scientific Diagram

The Control Centers of Biomolecular Phase Separation: How Membrane Surfaces, PTMs, and Active Processes Regulate Condensation - ScienceDirect

Condensation of Ded1p Promotes a Translational Switch from Housekeeping to Stress Protein Production - ScienceDirect

Systematic discovery of biomolecular condensate-specific protein phosphorylation | Nature Chemical Biology

Analysis of biomolecular condensates and protein phase separation with microfluidic technology - ScienceDirect

Protein condensation kinetic pathways crystallization and disease | Biological physics and soft matter physics | Cambridge University Press
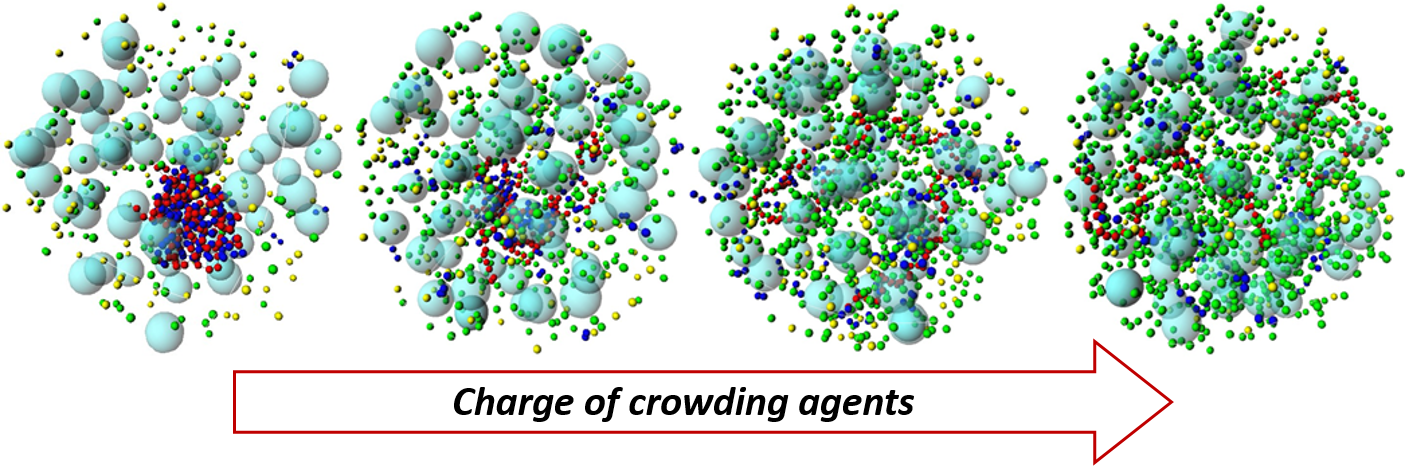
Polymers | Free Full-Text | Role of Protein Self-Association on DNA Condensation and Nucleoid Stability in a Bacterial Cell Model

![Natural Condensation Polymers [Proteins and Carbohydrates] (GCSE Chemistry) Natural Condensation Polymers [Proteins and Carbohydrates] (GCSE Chemistry)](https://i.ytimg.com/vi/LfCB2AEOuL0/maxresdefault.jpg)