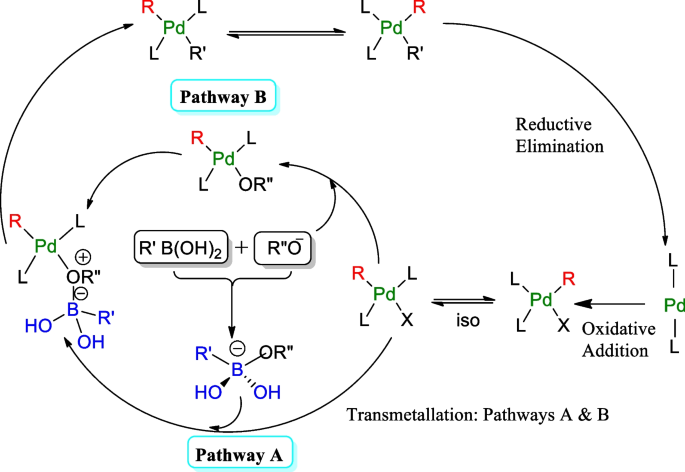
Suzuki–Miyaura cross-couplings for alkyl boron reagent: recent developments—a review | Future Journal of Pharmaceutical Sciences | Full Text

Pd-NHC catalyzed Suzuki cross-coupling of benzyl ammonium salts | Research on Chemical Intermediates

Scheme 1. Mechanism of the homogeneous Suzuki-Miyaura reaction. Scheme... | Download Scientific Diagram

Reasonable mechanism for the Suzuki–Miyaura cross coupling reaction of... | Download Scientific Diagram
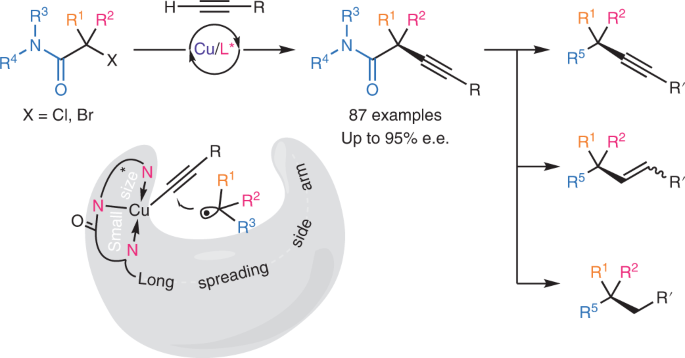
Mechanism-based ligand design for copper-catalysed enantioconvergent C(sp3)–C(sp) cross-coupling of tertiary electrophiles with alkynes | Nature Chemistry
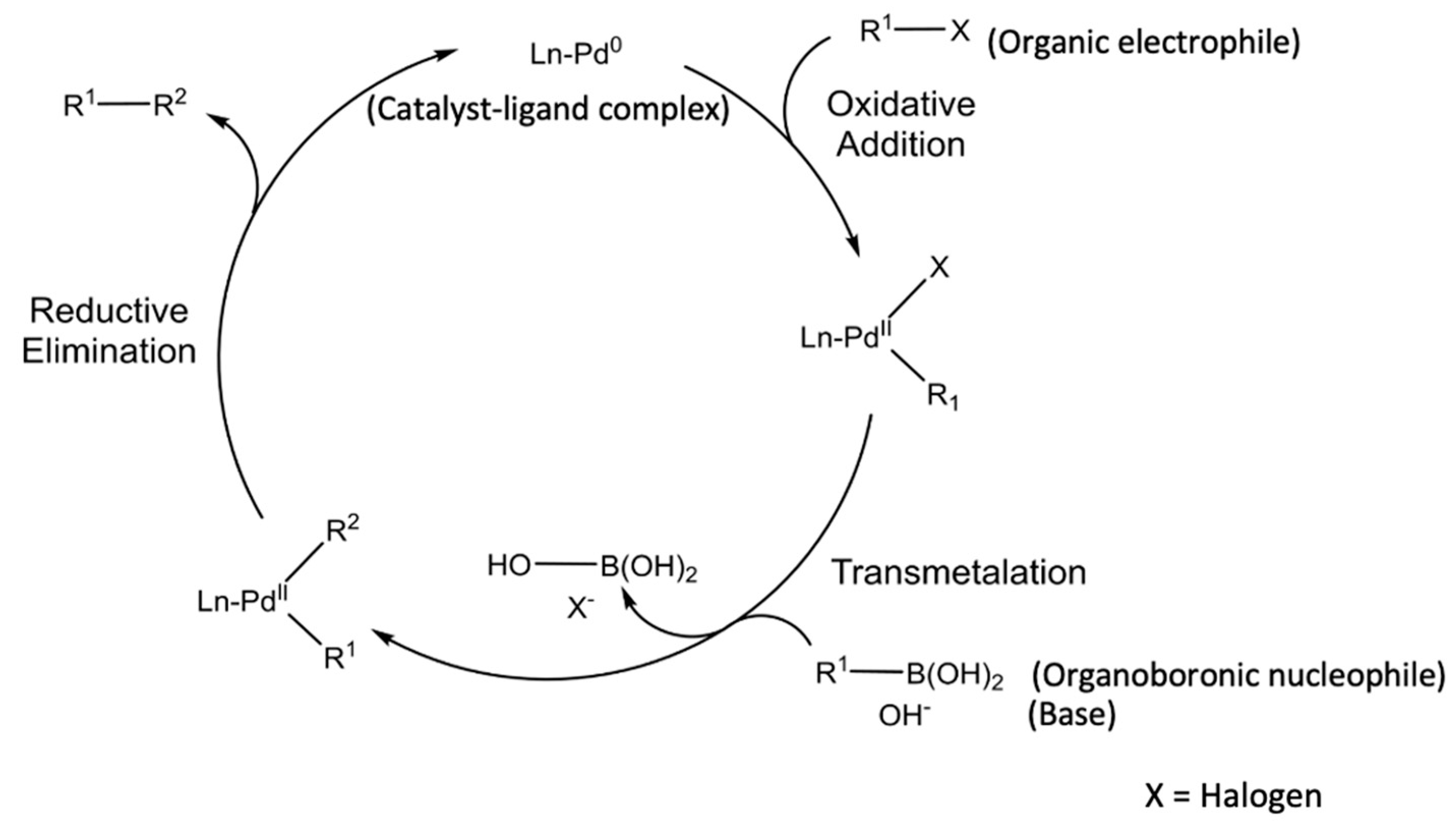
Knowledge | Free Full-Text | Catalyst Recycling in the Suzuki Coupling Reaction: Toward a Greener Synthesis in the Pharmaceutical Industry

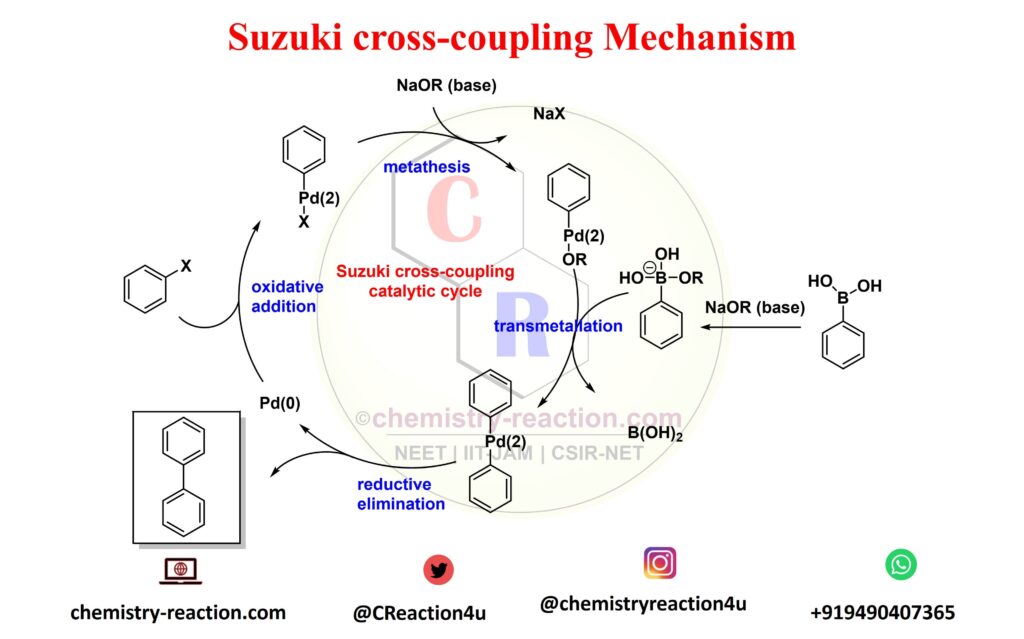


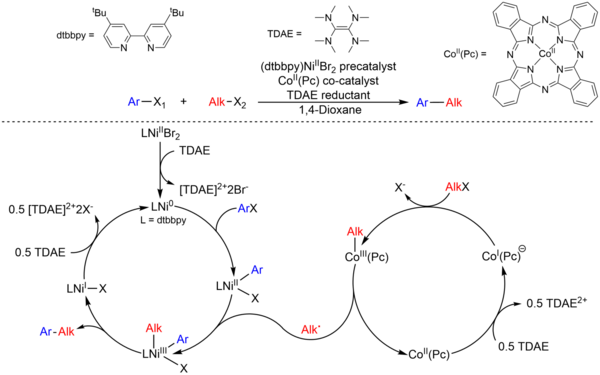

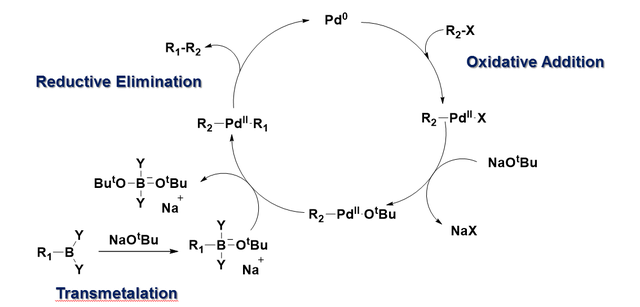

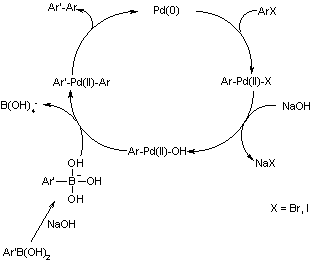

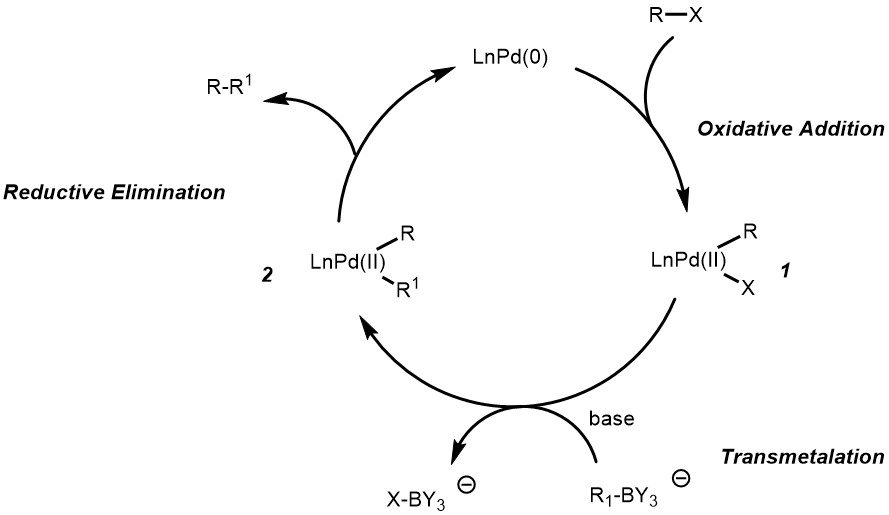
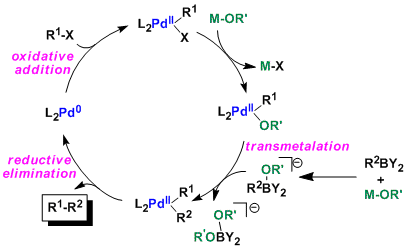
![46] Suzuki Cross Coupling 1979 – ChemInfoGraphic 46] Suzuki Cross Coupling 1979 – ChemInfoGraphic](https://cheminfographic.wordpress.com/wp-content/uploads/2017/11/46_suzuki_coupling1.jpg)

![PDF] Mechanisms of Nickel-Catalyzed Cross-Coupling Reactions | Semantic Scholar PDF] Mechanisms of Nickel-Catalyzed Cross-Coupling Reactions | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/3297cc29ee21e706a719bd5da81dca59c2ac6ab2/7-Figure4-1.png)
