The threshold frequency of a metal is 710 power 14 Hz.Calculate the kinetic energy of an electron emitted when radiation of frequency = 1 10 power 15 Hz hits the metal.

Calculate the threshold frequency of metal if the binding energy is `180.69 kJ mol^(-1)` of electron
the threshold frequency for a metal is 6.2×10.^3 8-1.calculate KE emitted of an electron when the radiation - Brainly.in
The threshold frequency v0 for a metal is 7.0 X 1014s 1. Calculate the kinetic energy of an electron emitted when radiation of frequency v = 1.0 X 1015 s 1 hits the metal.

Date: Date: 1 5 The threshold frequency of a metall is 16 X 10''s', Talculate the kinet's energy of a photo electron eninitted when radiatich o frequency 12 x18° 5' Do =

The photo-electric emission requires a threshold frequency nu_0. For a certain metal lambda_1=2200 A^{circ} and lambda_2=1900 A^{circ} produce electrons with a maximum kinetic energy KE_1 and KE_2. If KE_2 = 2KE_1, calculate

Cosmological Astrophysics - Threshold Frequency --- The minimum energy required to produce photo-emission of electrons is called the #work_function and this depends on the type of metal used. The minimum frequency of

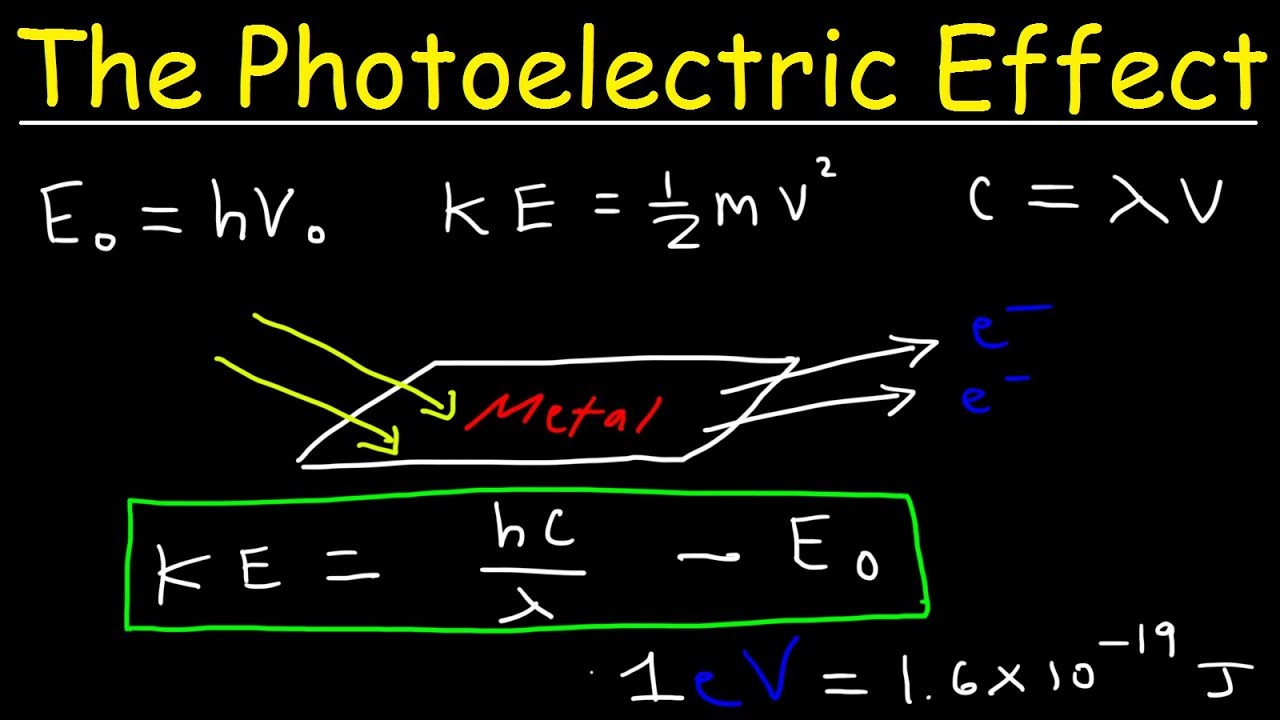
Photoelectric Effect, Work Function, Threshold Frequency, Wavelength, Speed & Kinetic Energy, Electr - YouTube
How To Calculate THRESHOLD Frequency For The Photoelectric Effect!! #Quantum #Mechanics #Physics #Chemistry #NicholasGKK

Finding the Frequency Threshold of Incident Light for Electron Emission from a Material with Given Work Function | Physics | Study.com