22. Calculate the entropy change when 2.8 g of N, gas expands isothermal e entopy change when 2.8 g of N, gas expands isothermally and reversibly from an initial volume of 1

PPT - Entropy Changes in the Surroundings Now that we have seen how to calculate D S system for reversible and irreversible PowerPoint Presentation - ID:28591

calculate the entropy change when 1 mole of an ideal gas is allowed to expand isothermally at 315k and pressure 5atm to 2.5atm - EduRev Class 11 Question
The entropy of vaporization of benzene is 85 JK^ 1mol^ 1. When 117 g benzene vaporizes at its normal boiling point then the entropy change of surrounding is : (1) 85 JK^ 1 (2) 85 × 1.5 JK^ 1 (3 ) 85 × 1.5 JK^ 1 (4) None of these .

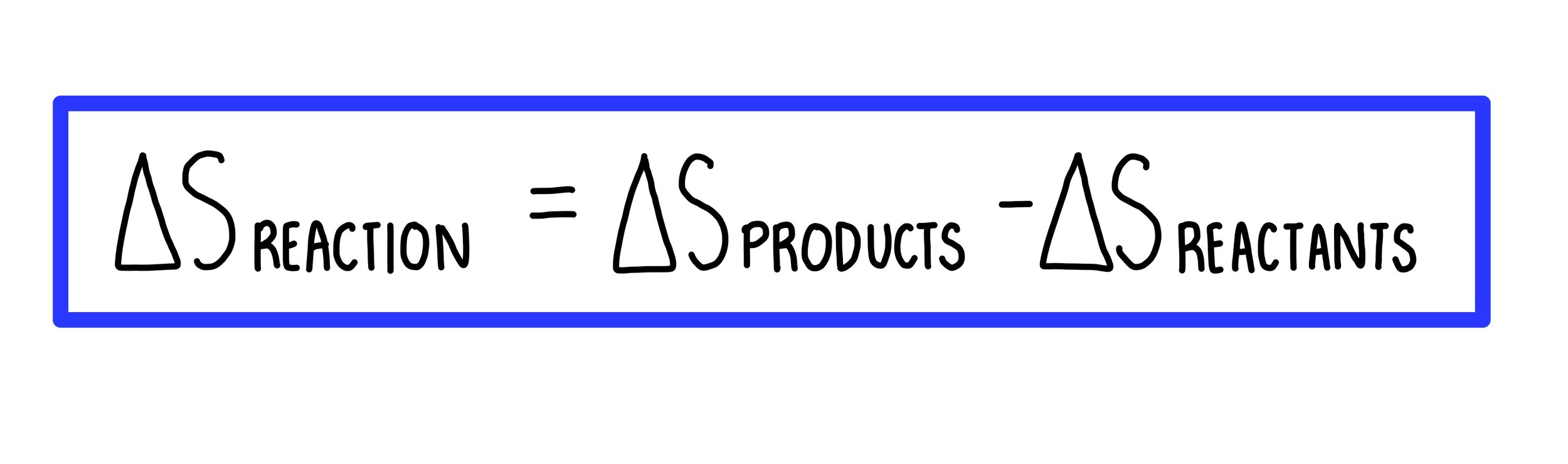
How to Calculate the Entropy Change for a Chemical or Physical Process Based on Absolute Entropies | Chemistry | Study.com

Calculate the entropy change accompanying conversion of 1 mole of ice at 273 K and 1 atm pressure into steam at 373 K and 1 atm pressure, given that at 273 K | Homework.Study.com
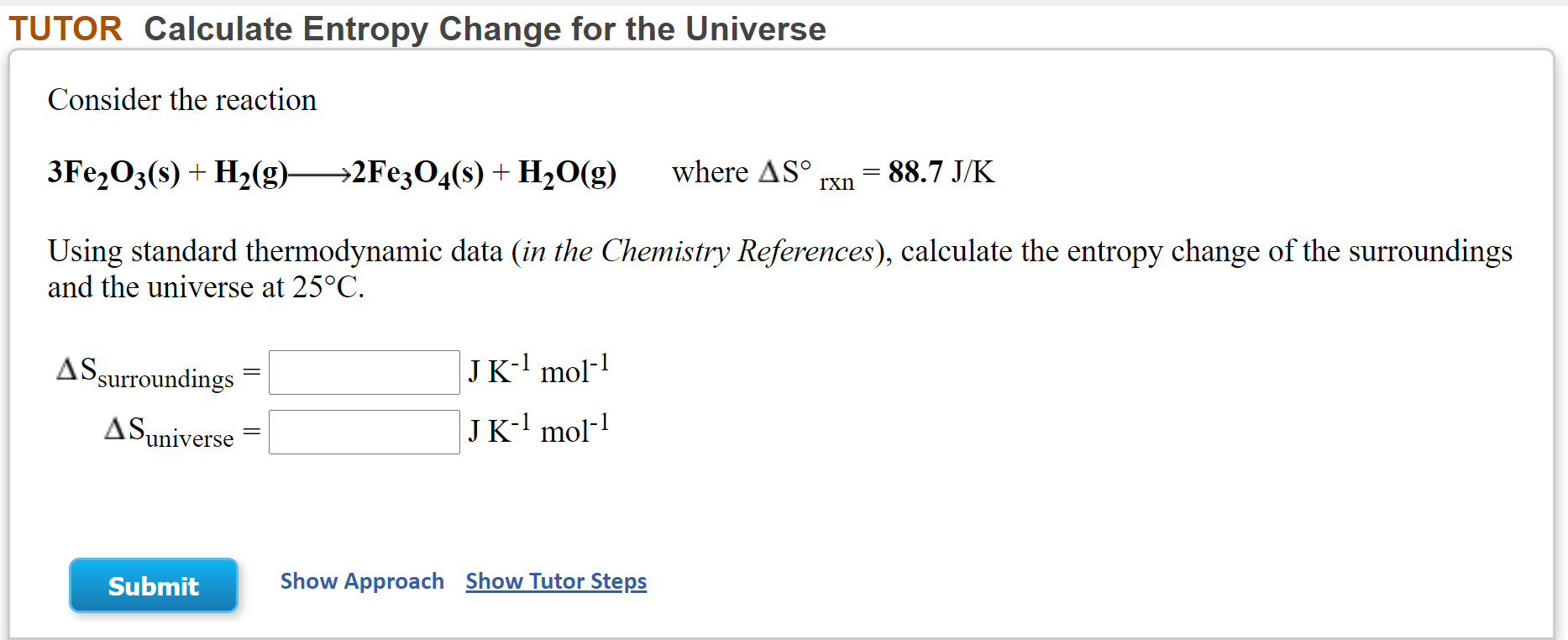
Calculate the entropy change in the system. and in the surroundings and the total entropy change in the universe when during - Sarthaks eConnect | Largest Online Education Community